Konference: 2015 20th Congress of the European Hematology Association - účast ČR
Kategorie: Myeloproliferativní nemoci
Téma: ePoster
Číslo abstraktu: E1415
Autoři: Prof. M.D. Georgi Mihaylov, Ph.D.; Galina Kuchma; MUDr. Jarmila Obernauerová; Dr. Barbara Skopec; MUDr. Zuzana Sninská; Nikolay Tzvetkov; Daniela Niepel; Peter Černelč
Background
Aims
The aim of this ongoing study is to assess the use of romiplostim in clinical practice in Bulgaria, Czech Republic, Slovenia, Slovakia, Romania and Russia.
Methods
This international single-arm, non-interventional, multicenter, retrospective and prospective study is enrolling adult pts with ITP, who have received at least one application of romiplostim according to the licensed indication. A period of 6 months prior to romiplostim initiation was documented retrospectively from pt files; the prospective observation period started at the time of romiplostim initiation. The total planned observation period is 2 years. All pts provided written informed consent. Assessed parameters included pt demographics, romiplostim application, dosage, adverse drug reactions (ADRs) and reasons of discontinuations, concomitant ITP therapies, clinically relevant bleeding events, and consumption of resources. Data from a protocol-defined interim analysis (IA) at 1 year of prospective observation are reported here.
Results
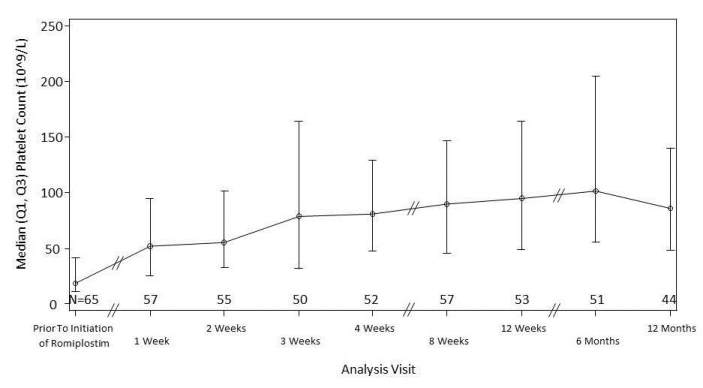
66 pts were analyzed for the IA (47% male, median age at romiplostim initiation 48 [range 19-82] years, splenectomized n=27, non-splenectomized n=39). Of these, 58 (88%) completed 1 year of study, 8 (12%) discontinued from the study (adverse drug reactions n=1, administrative decision n=2, lost to follow-up n=5). The median time from ITP diagnosis to romiplostim initiation was 2.89 (0-42) years. 34 pts (51.5%) had received ≥3 prior ITP therapies. During the 6 months before romiplostim initiation the event rate per 100 pt years was 111 (95% CI 75.1, 156.8) for bleeding and 92 (95% CI 73.2, 113.0) for ITP-related hospitalizations. The median platelet count at romiplostim start was 19.0 (range 1-411) x 10^9/L, increasing to 52.0 (range 2-361) x 10^9/L after 1 week and remaining above 50 x 10^9/L for the period until IA data cut-off at 1 year (Figure). During the 1-year observation period for IA, pts received a median romiplostim dose of 2.13 (range 0.1-9.1) µg/kg/week and a median of 31 (range 2-53) injections. The event rate per 100 pt years during the 1-year romiplostim treatment period was 58 (95% CI 39.0, 83.6) for bleeding (no grade 3/4 events) and 94 (95% CI 69.3, 125.4) for ITP-related hospitalizations. 4 pts (6.1%) experienced a total of 11 ADRs, with 2 pts experiencing 2 serious ADRs each (thrombosis, dysphagia, lymphocytosis, leucocytosis). 51 pts (77.3%) were hospitalized for 77 ITP-related events during the 1 year observation period for IA for the following reasons: platelet transfusion (n=5, 9.8%), ITP treatment administration (n=24, 47.1%), bleeding event (n=14, 27.5%), infection-related (n=5, 9.8%), splenectomy (n=7, 13.7%), ADR (n=1, 2.0%), other (n=21, 41.2%).
Summary
These interim data show that platelet counts in ITP patients treated with romiplostim in clinical practice increased and were maintained within the desired range of 50-250 x 10^9/L. The bleeding event rate was lower during romiplostim treatment. Romiplostim was generally well tolerated.
Keyword(s): Immune thrombocytopenia (ITP), Thrombocytopenia
Datum přednesení příspěvku: 12. 6. 2015
