Konference: 2008 XV. Jihočeské onkologické dny
Kategorie: Genitourinární nádory
Téma: Postery
Číslo abstraktu: 055p
Autoři: L. Zhang; prof. MUDr. Jiří Petera, Ph.D.; F. Orsi; W. Chen; H. Zhu; Z. Wang
BACKGROUND
As a non-invasive technique, ultrasound image-guided HIFU allows the very accurate destruction of solid tumors, without harming overlying and adjacent structures even within the path of the beam. The aim of this review was to demonstrate the clinical experience of using ultrasound image-guided HIFU to treat solid tumours.
HIFU THERAPEUTIC PROCEDURE
HIFU ablation was performed by the JC HIFU system [Chongqing Haifu (HIFU) Tech Co., Ltd., Chongqing, China]. Briefly, the therapeutic procedure was guided by real-time ultrasonography (US). Therapeutic US energy was produced by a transducer with a 15 cm diameter, a focal length of 15 cm, and operating at a frequency of 0.8 MHz. General anesthesia or concious sedation was necessary during HIFU treatment to prevent the patient from experiencing deep visceral-type pain and to ensure immobilization. After suitable anesthesia was induced, the patient was carefully positioned, so that the skin overlaying the lesion to be treated would be easily put in contact with the degassed water. The treatment power was increased stepwise after starting, and the ablation was terminated after the increased gray scale covered the tumour margin.
OVERVIEW
In 1942, Lynn et al1 reported for the first time use of HIFU to produce coagulative necrosis in target tissue. In 1950s, Fry et al2 conducted HIFU ablation of caťs brain tissue after removing a piece of skuli;
Over last decade, more than 10,000 of patients with uterine fibroids, liver cancers, breast cancer, pancreatic cancer, bone tumours, renal cancer have been treated with ultrasound image-guided HIFU. More than 50 centers in China, United Kindom, South Korea, Japan, Italy, Spain, Russia equipped with the JC HIFU system [Chongqing Haifu (HIFU) Tech Co., Ltd., Chongqing, China]. Briefly, the therapeutic procedure was guided by real-time ultrasonography (US), including centers at world-renowned institutes, such as Oxford Universty, European institute of Oncology (Milan, Italy), Chiba University (Japan), General Hospital of the People's Liberation Army (also Hospital 301, China), Queen Mary Hospital (Hong Kong)...
Recently, some research groups reported their results showing that HIFU ablation was safe, effective and feasible in treating human solid tumours345. The Group from Chongqing has reported the largest series of HIFU clinical applications to dáte, in which 1038 patients were treated with US guided HIFU in ten centers with both curative and palliative purposes6. Zhang et al.7 reported HIFU can achieve complete tumor necrosis even when the lesion is located adjacent to the major hepatic blood vessels. Indeed, there is no discernible damage to the major vessels, even though the adjacent tumor has been completely ablated. In Oxford, UK, a total of 22 patients with liver metastases were treated with USgHIFU. Using either radiological images such as MRI and contrast ultrasound, or histological examinations, 20 of 22 patients were assessed. The results revealed that the adverse event profile was favorable when compared to open or minimally invasive techniques8. This year, a feasiability study of ultrasonography imaging-guided high intensity focus ultrasound (HIFU) treatment for solid malignancies was completed in European Institute of Oncology (Milan, Italy)9; This will be the first report on HIFU treatment for solid malignant tumours from a European Comprehensive Cancer Center for defining clinical and technical indication for this technique.
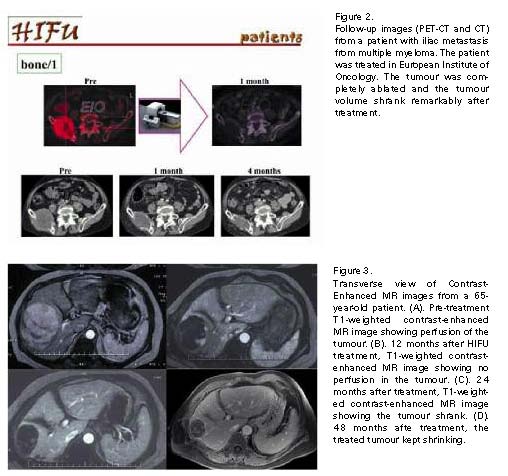
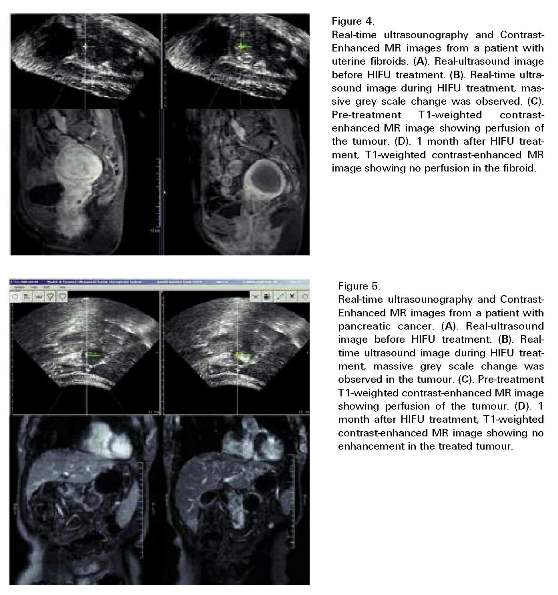
CONCLUSIONS
These results from different groups are exciting
and encouraging. Based on the results from clinical trials and
studies, we concluded US-guided HIFU ablation can achieve
completely response without significant side effects, therefore, it
is a safe, effective and feasible modality for the destruction of
malignant solid tumours.
Reference
- Lynn JG, Zwemer RL, Chick AJ, et al. A new method for the
generation and use of focused US in experimental biology. J Gen
Physiol 1942; 26:179-193.
- Fry WJ, Barnard JW, Fry FJ. Ultrasonically produced localized
selective lesions in the central nervous system. AmJPhysMed
1955;34:413-423.
- Hynynen K, Pomeroy O, Smith DN, et al. MR imaging-guided
focused ultrasound surgery of fibroadenomas in the breast: A
feasibility study. Radiology 2001; 219:176-185.
- Wu F, Wang ZB, Cao YD, et al. Changes in biologie
characteristics of breast cancer treated with high-intensity
focused ultrasound. Ultrasound Med Biol 2003;
29:1487-1492.
- Feng Wu, Zhi-Biao Wang, Hui Zhu, Wen-Zhi Chen, Jian-Zhong Zou,
Jin Bai, Ke-Quan Li, Cheng-Bing Jin, Fang-Lin Xie, Hai-Bing Su,
Feasibility of US-guided High-lntensity Focused Ultrasound
Treatment in Patients with Advanced Pancreatic Cancer: Initial
Experience. Radiology 2005; 236:1034-1040.
- Wu F, Wang ZB, Chen WZ, Wang W, Gui Y, Zhang M, Zheng G, Zhou
Y, Xu G, Li M, Zhang C, Ye H, and Feng R. Extracorporeal high
intensity focused ultrasound ablation in the treatment of 1038
patients with solid carcinomas in China: an overview. Ultrason
Sonochem 2004; 11:149-154.
- Lian Zhang, Hui Zhu, Chengbin Jin, Kun Zhou, Kequan Li, Haibing
Su, Wenzhi Chen, Jin Bai, Zhibiao Wang (2008) High intensity
focused ultrasound (HIFU): Effective and safe therapy for
hepatocellular carcinoma adjacent to major hepatic veins. Eur
Radiol: accepted
- RO llling, JE Kennedy, F Wu, GR ter Haar, AS Protheroe, PJ
Friend, FV Gleeson, DW Cranston, RR Phillips and MR Middleton. The
safety and feasibility of extracorporeal high-intensity focused
ultrasound (HIFU) for the treatment of liver and kidney tumours in
a Western population. British Journal of Cancer (2005) 93,
890-895.
- Franco Orsi, Lian Zhang, Paolo Arnone, et al. Feasiability of ultrasonography imaging-guided high intensity focus ultrasound (HIFU) treatment for solid malignancies in a European Comprehensive Cancer Center, (in submitting)
Datum přednesení příspěvku: 10. 10. 2008
