Konference: 2015 57th ASH Annual Meeting - účast ČR
Kategorie: Maligní lymfomy a leukémie
Téma: 624. Lymphoma: Therapy with Biologic Agents, excluding Pre-Clinical Models: Biologic Agents in B Cell Lymphoma
Číslo abstraktu: 469
Autoři: prof. Simon A. Rule; Wojciech Jurczak; MD Mats Jerkeman, Ph.D.; Dr. Rodrigo Santucci Silva; MD Chiara Rusconi; Prof. MUDr. Marek Trněný, CSc.; MD Fritz C. Offner, PhD; MD Dolores Caballero; MD Cristina Joao, Ph.D.; MD Mathias Witzens-Harig; MD Georg Hess; Isabelle Bence-Bruckler; MD Seok-Goo Cho; Sriram Balasubramanian, Ph.D.; Nibedita Bandyopadhyay, Ph.D.; Steven Sun; MD Jenna Goldberg; John Bothos, Ph.D.; Christopher Enny; Shana Traina, Ph.D.; MD Aleksandra Rizo; MD Jessica Vermeulen, Ph.D.; MD Martin H. Dreyling, Ph.D.
MCL is an aggressive B-cell lymphoma with a poor overall prognosis. For patients who fail initial therapy, conventional chemotherapy achieves only short-term remissions. Ibrutinib is a first-in-class, once-daily, oral, covalent inhibitor of Bruton’s tyrosine kinase that has been shown to be highly active for previously treated MCL patients (overall response rate [ORR] ~65%; complete response [CR] ~20%) in single-arm phase 2 studies. Temsirolimus has demonstrated significantly longer progression-free survival (PFS) vs investigator’s choice. In this phase 3, randomized, open-label study (MCL3001 [RAY]), ibrutinib was compared with temsirolimus in patients with relapsed or refractory (R/R) MCL who had received ≥1 prior rituximab-containing therapy.
Methods
Patients were randomized at a 1:1 ratio to receive oral ibrutinib (560 mg once-daily) or intravenous temsirolimus (175 mg: Days 1, 8, and 15 of Cycle 1; 75 mg: Days 1, 8, and 15 of subsequent cycles). Stratification factors were number of prior lines of therapy (1-2 vs ³ 3) and simplified MCL international prognostic index (sMIPI) risk (low [0-3] vs intermediate [4-5] vs high [6-11]). Patients in both arms received treatment until disease progression or unacceptable toxicity. Primary end point of the study was PFS, as assessed by an independent review committee (IRC). Secondary end points included ORR, overall survival (OS), time to next treatment (TTNT), time to worsening of lymphoma symptoms (measured by FACT-Lym lymphoma subscale), and safety.
Results
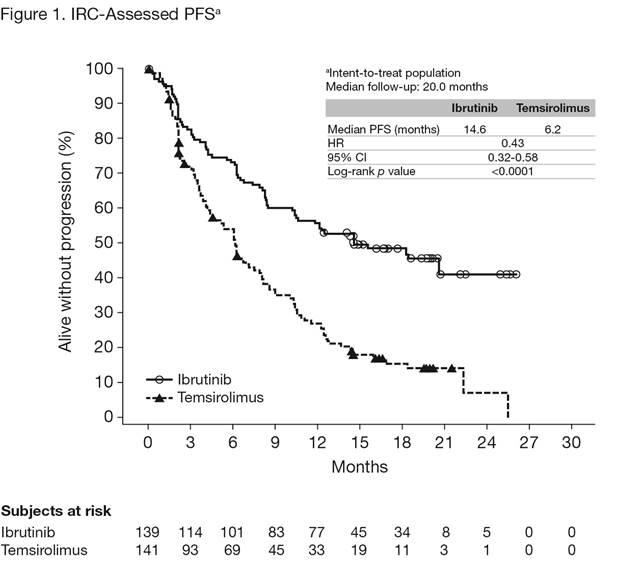
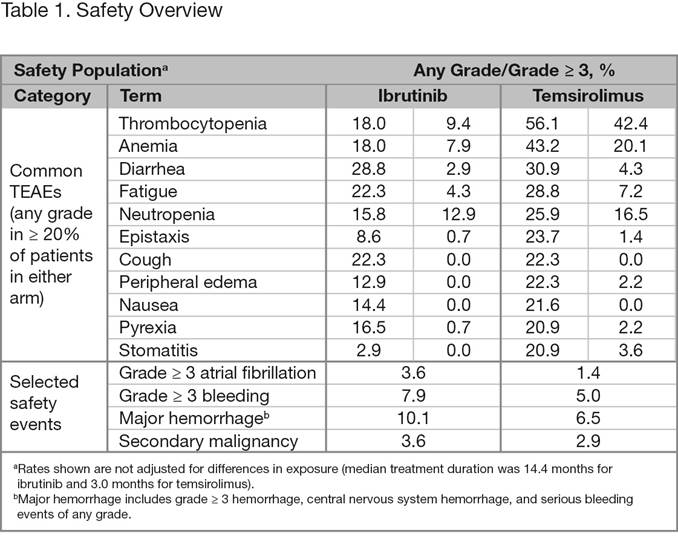
Overall, 280 patients were randomized to ibrutinib (n = 139) or temsirolimus (n = 141). Baseline disease characteristics and demographics were generally well balanced. Median age (range) was 68 years (34-88) and median number (range) of prior lines of therapy was 2 (1-9). Approximately two-thirds of the patients had intermediate- or high-risk disease. At the time of this analysis, median follow-up was 20.0 months. Ibrutinib was superior to temsirolimus for the primary end point of IRC-assessed PFS, with a statistically significant 57% reduction in the risk of progression or death (Figure 1). The median PFS was 14.6 months for the ibrutinib arm and 6.2 months for the temsirolimus arm, respectively. At a landmark of 2 years, the PFS rate is 41% in the ibrutinib arm vs 7% in the temsirolimus arm. PFS results were consistent across most assessed subgroups. Investigator-assessed PFS was consistent with the IRC results. IRC-assessed ORR was significantly higher for ibrutinib vs temsirolimus (71.9% vs 40.4%; p < 0.0001) with a CR rate of 18.7% vs 1.4%, respectively. Median OS was not reached with ibrutinib vs 21.3 months with temsirolimus, showing a positive trend toward patients in the ibrutinib arm (reduced risk of death by 24% [HR, 0.76; 95% CI, 0.53-1.09]). However, these results may have been confounded by 23% of patients that initially received temsirolimus crossing over to receive ibrutinib. A greater proportion of patients treated with ibrutinib vs temsirolimus avoided worsening of lymphoma symptoms throughout the study; 27% of ibrutinib patients had worsening vs 52% of temsirolimus patients. Median TTNT was not reached with ibrutinib vs 11.6 months with temsirolimus. Median treatment duration was 14.4 months for ibrutinib and 3.0 months for temsirolimus. Overall, 6.5% of subjects discontinued treatment due to AEs in the ibrutinib arm and 25.5% of subjects discontinued treatment due to AEs in the temsirolimus arm. The most common TEAEs with ibrutinib were diarrhea, fatigue, and cough, whereas with temsirolimus, thrombocytopenia, anemia, and diarrhea were most commonly observed (Table 1). Grade ³ 3 TEAEs were reported for 67.6% of ibrutinib patients vs 87.1% of temsirolimus patients; most frequent were thrombocytopenia, anemia, and neutropenia. When adjusted for exposure, TEAE incidence was consistently lower for the ibrutinib arm vs the temsirolimus arm.
Conclusions
Ibrutinib is superior to temsirolimus for PFS and ORR, and showed preferable tolerability. The results of this phase 3 trial confirm the efficacy and favorable safety profile of ibrutinib shown in phase 2 studies. Future concepts will investigate ibrutinib-based combination approaches for patients with R/R MCL.
Disclosures: Rule: Gilead: Research Funding ; Celgene: Consultancy , Other: Travel reimbursement ; J&J:Consultancy , Other: Travel reimbursement , Research Funding ; Roche: Consultancy , Other: Travel reimbursement . Rusconi: Roche: Honoraria . Joao: Celgene, Novartis: Consultancy ; Janssen, Roche: Membership on an entity’s Board of Directors or advisory committees . Witzens-Harig: Pfizer: Honoraria , Research Funding ;Roche: Honoraria . Hess: Pfizer, Janssen, Roche, Mundipharma: Honoraria , Research Funding ; Janssen, Roche, , Celgene, Novartis: Consultancy . Balasubramanian: Janssen: Employment , Equity Ownership ; Pharmacyclics LLC, an AbbVie Company: Equity Ownership . Bandyopadhyay: Janssen: Employment . Sun: Janssen/J&J: Employment , Equity Ownership . Goldberg: Janssen: Employment . Bothos: Janssen: Employment . Enny: Janssen: Employment .Traina: Janssen: Employment . Rizo: Janssen: Employment . Vermeulen: Janssen: Employment .
Datum přednesení příspěvku: 7. 12. 2015