Konference: 2014 19th Congress of the European Hematology Association - účast ČR
Kategorie: Mnohočetný myelom
Téma: Myeloma and other monoclonal gammopathies - Clinical
Číslo abstraktu: S1338
Autoři: M.D. Federica Cavallo; MD Andrew Spencer; Francesca Gay; prof. MUDr. Roman Hájek, CSc.; MD Maria Teresa Petrucci; M.D. Dina Ben-Yehuda; M.D. Angelo Michele Carella; Letizia Maria Vallone; Stefania Oliva; Daniela Petrò; Claudia Crippa; Dr. Agostina Siniscalchi; Milena Gilestro; prof. MUDr. Luděk Pour, Ph.D.; MD Laura Maracci; Norbert Pescosta; Prof. MD Anna Marina Liberati; MD Valeria Magarotto; Prof.MUDr. Vlastimil Ščudla, CSc.; Giulia Benevolo; M.D. Stefano Pulini; MD Tommaso Caravita; Izhar Hardan; MD Mario Boccadoro; MD Antonio P. Palumbo
ABSSUB-4452
Background: Autologous stem cell transplantation (ASCT) improved outcome compared to conventional chemotherapy in newly diagnosed multiple myeloma (NDMM) patients. The role and timing of ASCT in the era of novel agents is a crucial question. PFS1 defines the time from start of therapy until the occurrence of 1st relapse. PFS2 defines the time from start of therapy until the occurrence of 2nd relapse, incorporating the duration of both 1st and 2nd remission.
Aims: To compare early ASCT versus ASCT at relapse in terms of PFS1, PFS2 and OS.
Methods: We analyzed pooled data from two phase III multicenter randomized trials including NDMM patients younger than 65 years (RVMM209: lenalidomide-based induction followed by ASCT vs chemotherapy plus lenalidomide consolidation and subsequent lenalidomide maintenance vs no maintenance; RVMMEMN441:lenalidomide-based induction followed by ASCT vs chemotherapy plus lenalidomide consolidation and subsequent lenalidomide-dexamethasone vs lenalidomide alone maintenance). In both trials, patients randomized to chemotherapy plus lenalidomide consolidation arm (CC group) who experienced progressive disease during treatment were allowed to receive ASCT at relapse. Patients randomized to ASCT consolidation arm (ASCT group) received treatment at relapse at the physician’s discretion. We evaluated PFS1 (time from start of consolidation to 1st relapse), PFS2 (time from start of consolidation to 2nd relapse), and OS (time from randomization at diagnosis to death) through a stratified analysis by protocol. At 1st relapse, 2nd PFS (time from 1st relapse to 2nd relapse) and survival from relapse (time from 1st relapse to death) were evaluated. Intention to treat analysis was performed including patients who were eligible for consolidation.
A subgroup analysis of PFS1, PFS2 and OS in patients who received early ASCT vs ASCT at relapse according to age, gender, protocol, ISS stage, cytogenetic profile and maintenance regimen was conducted.
Results: A total of 791 patients were enrolled in the two trials and 529 were eligible for consolidation: 268 in the ASCT group and 261 in the CC group. Baseline characteristic were equally distributed in the two groups. Median follow-up for survivors was 44.5 months. Early ASCT significantly improved PFS1 (3-year rate: 59% vs 35%, HR 0.48, CI 95% 0.37-0.62, P<0.001) and PFS2 (3-year rate: 77% vs 68%, HR 0.59, CI 95% 0.39-0.89, P=0.012), and marginally OS (4-year rate: 83% vs 72%, HR 0.64, CI 95% 0.38-1.08, P=0.096) in comparison with ASCT at relapse (Figure). 99 patients in the ASCT group experienced 1st relapsed in comparison with 159 patients in the CC group. 42% of patients in the CC group did not receive ASCT at relapse. No differences in 2nd PFS and OS from relapse between patients in the ASCT group or the CC group was noticed. The advantage of early ASCT was also observed when the two trials were analyzed separately and was confirmed in the subgroup analysis.
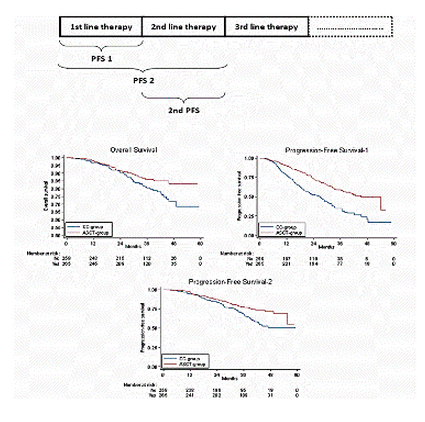
Summary/Conclusion: In this pooled analysis, early ASCT improved PFS1, PFS2 and OS in NDMM patients. These results confirm the importance of ASCT as 1st line therapy even in the era of novel agents.
Keywords: Autologous hematopoietic stem cell transplantation, Multiple myeloma, Young adult
Datum přednesení příspěvku: 15. 6. 2014
