Konference: 2015 20th Congress of the European Hematology Association - účast ČR
Kategorie: Maligní lymfomy a leukémie
Téma: Poster
Číslo abstraktu: P209
Autoři: Panagiotis Baliakas; Anastasia Hadzidimitriou; Dr. Mattias Mattsson; Aliki Xochelli; Lesley-Ann Sutton; Evangelia Minga; Lydia Scarfo; M.D. Davide Rossi; Zadie Davis; Andreas Agathangelidis; M.D. Neus Villamor, Ph.D.; Helen Parker, Ph.D.; Mgr. Jana Kotašková, Ph.D.; Evangelia Stalika; Mgr. Karla Plevová, Ph.D.; Larry Mansouri; Diego Cortese; Alba Navarro (Lopez), Ph.D.; M.D. Julio Delgado, Ph.D.; Dr. Marta Larrayoz; Dr. Achilles Anagnostopoulos; MD Chrysoula Belessi; M.D. Karin Ekström Smedby, Ph.D.; Prof. MD Gunnar Juliusson, PhD; BSc. Jonathan C. Strefford, PhD; prof. RNDr. Šárka Pospíšilová, Ph.D.; Prof. David Graham Oscier; MD Gianluca Gaidano, PhD; M.D. Elias Campo, Ph.D.; M.D. Paolo Ghia, Ph.D.; Prof. MD Richard Rosenquist (Brandell), PhD; MD Kostas Stamatopoulos
Background
Chronic lymphocytic leukemia (CLL) patients with somatically
hypermutated IGHV genes (M-CLL) within their B-cell receptor
immunoglobulin (BcR IG) generally display favorable biological
profiles with an indolent disease course. However, neither the
biological background nor the clinical presentation and outcome of
M-CLL is uniform, thus justifying the quest for parameters that
would assist in refining prognosis.
Aims
Development of a novel prognostic index tailored to M-CLL.
Methods
Cox-regression models were used to assess the impact of
demographic, clinical, genomic and immunogenetic features on
time-to-first-treatment (TTFT) in 1359 M-CLL cases.
Results
Main features of the studied cohort were as follows: median age: 64
years (22-92); males: 810/1359 (60%), Binet A/B/C: 1169/79/53
(90%/6%/4%); CD38 expression (CD38+): 161/1225 (13%). Except for
del(13q) (494/836, 59%) and trisomy 12 (+12, 142/1072, 13%), other
genomic aberrations were relatively rare [del(11q): 49/1076 (4.5%),
TP53 abnormality (TP53abn) i.e. del(17p) and/or
TP53 mutations: 52/1144 (4.5%), NOTCH1 mutations
(NOTCH1m): 23/1333 (2%), SF3B1m: 33/897 (4%),
MYD88m: 23/556 (4%)]. Borderline mutated IGHV genes
(germline identity, GI: 97-97.99%) were expressed by 101 cases
(7%). Thirty-three (2.5%) and 26 (2%) patients were assigned to
stereotyped subsets #4 (IGHV4-34/IGKV2-30) and #2
(IGHV3-21/IGLV3-21), respectively. All the aforementioned
parameters were evaluated for their impact on TTFT, using
univariate regression analysis. As expected, Binet stage C at
diagnosis emerged as the most powerful marker (HR: 9.44, 95% CI,
7.45-11.97, p<0.0001). Among the remaining cases (Binet A/B),
representing 96% of the cohort, the following parameters were
significantly associated with shorter TTFT: Binet B (HR: 7.35,
5.51-9.81, p<0.0001), subset #2 membership (HR: 4.24, 2.32-7.76,
p=0.0001), CD38+ (HR: 2.62, 1.98-3.50, p<0.0001),
TP53abn (HR: 2.56, 1.64-4, p=0.0003), +12 (HR: 1.75,
1.27-2.41, p=0.002), male gender (HR: 1.52, 1.2-1.9, p=0.004). In
contrast, a trend for favorable outcome was observed for subset #4
membership (HR: 0.43, 0.16-1.18, p=0.06). Taking into consideration
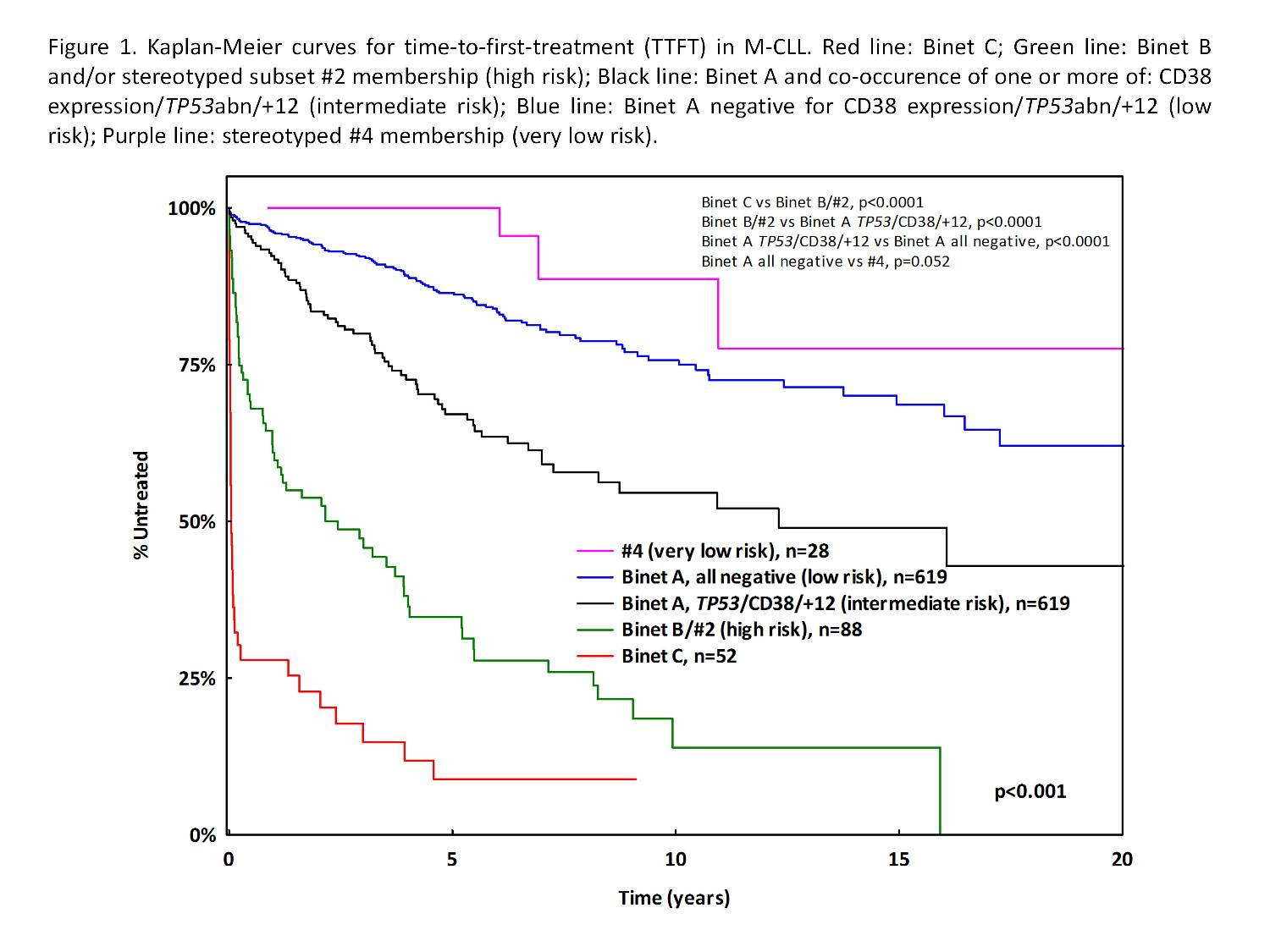
the above HRs as well as median TTFT, we defined 4 groups among
Binet A/B patients (n=934) with different TTFT (Figure 1): (i) high
risk (n=88, 9%): Binet B or subset #2 membership (median TTFT: 2.1
years, 65% and 85% probability of treatment initiation (TI) at 5
and 10 years from diagnosis, respectively); (ii) intermediate risk
(n=199, 22%): Binet A and occurence of one or more of: CD38
expression/TP53abn/+12 (median TTFT: 12 years, 33% and 45%
TI at 5 and 10 years, respectively); (iii) low risk (n=619, 66%):
Binet A with no CD38+/TP53abn/+12 (median TTFT: not yet
reached, 15% and 25% TI at 5 and 10 years, respectively); and (iv)
very low risk (n=28, 3%): subset #4 membership (median TTFT not yet
reached, 0% and 10% TI at 5 and 10 years,
respectively).
Summary
We report a simple prognostic index for M-CLL capable of identifying patients who will experience short TTFT as well as those who will probably never require treatment, applicable even within early clinical stage CLL. At the ends of the clinical spectrum described by this novel index lie stereotyped subsets #2 (aggressive) and #4 (indolent). This further highlights the importance of distinct BcR IG configuration, likely linked to distinct immune signaling, for CLL ontogeny and evolution and indicates that subclassifying M-CLL based on stereotypy is relevant in the context of everyday routine practice.
Keyword(s): Chronic lymphocytic leukemia,
Prognosis
Datum přednesení příspěvku: 22. 6. 2015
